NucleoMag Dx Pathogen, CE-IVD certified kit for isolation of viral RNA
*taxes and shipping not included
Delivery time approx. 5 working days
Magnetic bead based kit for isolation of viral RNA from oral/nasal swabs and saliva (validated für SARS-CoV-2; compliant with IVDR 2017/746)
| Application | Isolation of viral RNA |
| Target | Viral RNA |
| CE certified | Yes, CE-IVD labelled for diagnostic use in the EU and other countries |
| Availability | Für US-Kunden bestellen Sie bitte das äquivalente NucleoMag Pathogen, REF 744210.4 (nur für Forschungszwecke) |
| Technology | Magnetic bead technology |
| Brand | NucleoMag |
| Format | Magnetic beads |
| Handling | Magnetic separation |
| Automated use | Yes |
| Automation support | Compatible with most open robotic platforms. Please visit www.mn-net.com/automation or consult our technical support for further information. |
| Sample material | Respiratory swabs, saliva (human) |
| Sample amount | ≤ 200 µL swab wash solution/saliva |
| Fragment size | 300 bp–approx. 50 kbp |
| Typical recovery | Depending on sample amount and quality |
| Theoretical binding capacity | 0.4 µg/µL beads |
| Elution volume | 50–100 µL |
| Preparation time | 40-120 min/96 preps, depending on instruments/automation used |
| Typical downstream application | NGS, RT−PCR |
| Storage temperature | 18–25 °C / 64–77 °F |
| Shelf life (from production) | 19 Month(s) |
| Gross weight (incl. packaging) | 1897 g / 4.18 lbs |
| Packaging dimensions | 220 x 220 x 234 mm / 8.66 x 8.66 x 9.21 Inch |
| Hazardous material | Yes |
- Download Instruction NucleoMag Dx Pathogen
- Download Instruction NucleoMag Dx Pathogen (DE)
- Download Instruction NucleoMag Dx Pathogen (FR)
- Download Instruction NucleoMag Dx Pathogen (ES)
- Download NucleoMag Dx Pathogen Script support
- Download Flyer NucleoMag Dx Pathogen
- Download Instruction NucleoMag Dx Pathogen (CZ)
- Download Instruction NucleoMag Dx Pathogen (HU)
- Download Instruction NucleoMag Dx Pathogen (IT)
- Download Instruction NucleoMag Dx Pathogen (PL)
NucleoMag Dx Pathogen*
*Not for sale in the US. Please inquire.
The NucleoMag Dx Pathogen kit is intended for the isolation of viral RNA from human respiratory samples, including nasal and oral swabs as well as saliva. The kit includes ready to use buffers, Proteinase K and Carrier RNA. The purification of viral RNA is based on the reversible binding of nucleic acids to the NucleoMag magnetic beads.
The kit has been validated for SARS-CoV-2 specific diagnostic workflows and shows a market leading performance enabling reliable detection of RNA viruses from clinical samples. The kit can be combined with sample collection devices and downstream assays of your choice**. Components and protocol of the NucleoMag Dx Pathogen kit are identical to the kit for research purposes. However, the intended use is limited to viral RNA from swabs and saliva.
** It is the sole responsibility of the user to validate the performance in combination with a particular downstream assay and / or automation device.
- CE-IVD-certified for viral RNA isolation for use with human respiratory swabs and saliva
- Magnetic bead based purification with established automation workflows** on various open access platforms
- excellent diagnostic sensitivtiy
Application data

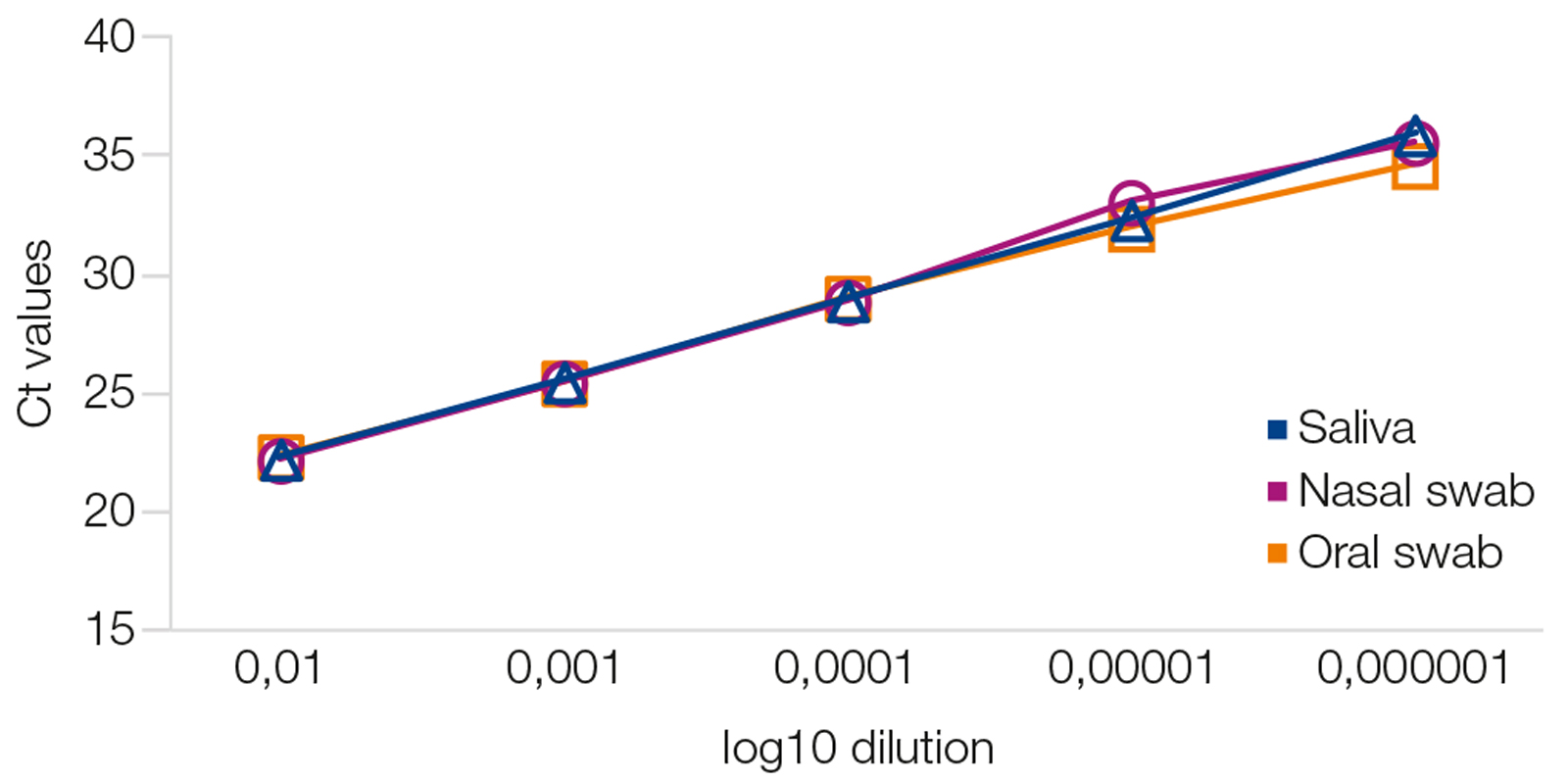
Consistent performance with different sample types
A dilution series of inactivated SARS-CoV-2 viruses was created in three different sample types (nasal swabs, oral swabs, saliva). RNA was extracted using the NucleoMag Dx Pathogen on a KingFisher™ Flex system. Viral RNA was quantified via specific qRT-PCR (AgPath ID™ One Step RT PCR mix + nCoV IP4 assay, Institute Pasteur, Paris). Viral RNA was detected consistently and reliably over a a range of five log10 dilutions.
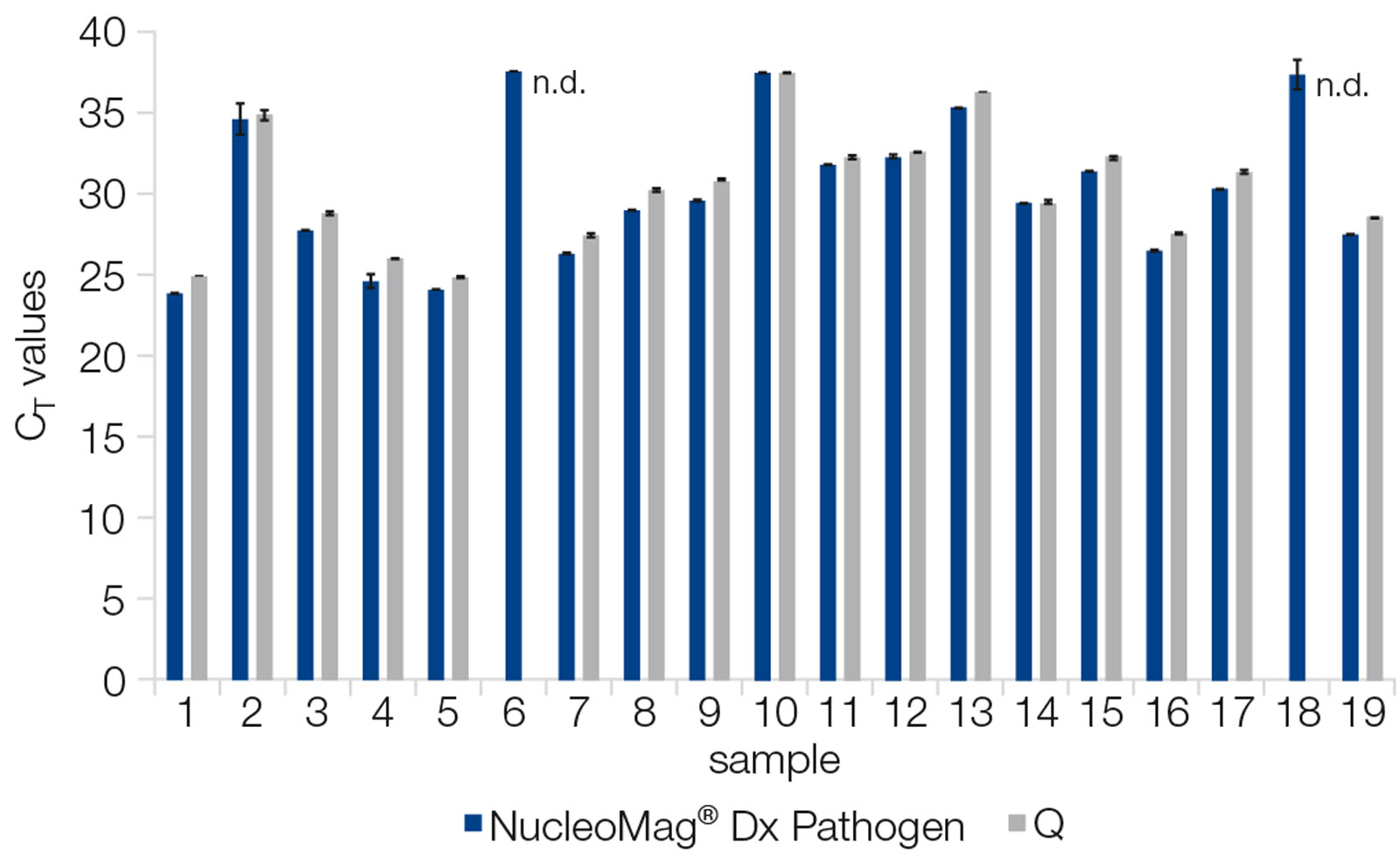
Excellent diagnostic sensitivity – competitor comparison
Viral RNA from 19 SARS-CoV-2 positive samples (duplicates) was extracted with the NucleoMag Dx Pathogen on a KingFisher™ Flex and a competitor kit (Q). Viral RNA was quantified with a SARS-CoV-2 specific qRT-PCR assay (qScript XLT One Step RT qPCR ToughMix + nCoV IP4 assay; Institute Pasteur, Paris). The NucleoMag Dx Pathogen kit performed equally well (3/19) or better (14/19) than the competitor kit. For two samples extracted with Q the CT could not be determined (n.d.).
Script support for automation


Basic scripts or protocols for the implementation of the NucleoMag Dx Pathogen kit on different automation platforms can be provided by MACHEREY-NAGEL.
If the NucleoMag Dx Pathogen kit is used in in-vitro diagnostic workflows, the use of the kit must be validated by the user in conjunction with the automation platform and consumables used and the subsequent in-vitro diagnostic assay (e.g., qRT-PCR). It is recommended to use appropriate controls (e.g. internal controls, extraction controls, positive / negative controls).
Individual protocol steps may vary depending on available consumables, hardware, platform and instrument setup.
Click here to read about the MACHEREY-NAGEL script support for NucleoMag Dx Pathogen.
 NucleoMag Pathogen kit for viral/bacterial DNA and viral RNA from clinical spl.
NucleoMag Pathogen kit for viral/bacterial DNA and viral RNA from clinical spl.  NucleoSpin Dx Virus, Mini kit for CE certified purification of viral RNA/DNA
NucleoSpin Dx Virus, Mini kit for CE certified purification of viral RNA/DNA 






