NucleoSpin Dx Blood, CE certified Mini kit for DNA from blood
*taxes and shipping not included
Mini spin kit for the isolation of DNA from blood (compliant with IVDR 2017/746)
| Application | Isolation of DNA |
| Selling unit | 50 Prep(s), 250 Prep(s) |
| Target | DNA |
| CE certified | Yes, CE-IVD labelled for diagnostic use in the EU and other countries |
| Availability | Für US-Kunden bestellen Sie bitte das äquivalente NucleoSpin Blood, REF 740951.50 |
| Technology | Silica membrane technology |
| Brand | NucleoSpin |
| Format | Mini prep |
| Handling | Centrifugation |
| Automated use | No |
| Sample material | Blood |
| Sample amount | 200 µL |
| Typical yield | 3–5 µg (depending on individual blood sample) |
| Typical concentration | 40–60 ng/µL |
| Typical purity A260/A280 | 1.9–2.1 |
| Elution volume | 50–200 µL |
| Preparation time | 30 min/prep |
| Typical downstream application | CE-IVD marked kit, DNA to be used with any downstream application employing enzymatic amplification |
| Storage temperature | 18–25 °C / 64–77 °F |
| Shelf life (from production) | 24 Month(s) |
| Hazardous material | Yes |
- Download Instruction NucleoSpin Dx Blood
- Download Instruction NucleoSpin Dx Blood (DE)
- Download Instruction NucleoSpin Dx Blood (FR)
- Download Instruction NucleoSpin Dx Blood (PL)
- Download Instruction NucleoSpin Dx Blood (NL)
- Download Instruction NucleoSpin Dx Blood (ES)
- Download Instruction NucleoSpin Dx Blood (EL)
- Download Instruction NucleoSpin Dx Blood (RO)
- Download Instruction NucleoSpin Dx Blood (HU)
- Download Instruction NucleoSpin Dx Blood (CZ)
NucleoSpin DX Blood*
*Not for sale in the US. Please inquire.
- CE-IVD marked in accordance with EU Directive 98/79/EC
- Fits into in vitro diagnostic workflows*
- Suitable for EDTA, citrate, and heparin blood from common blood collecting systems
- For fresh and frozen blood samples
- Reproducible results for reliable downstream applications
- Available as 50 prep and 250 prep kit
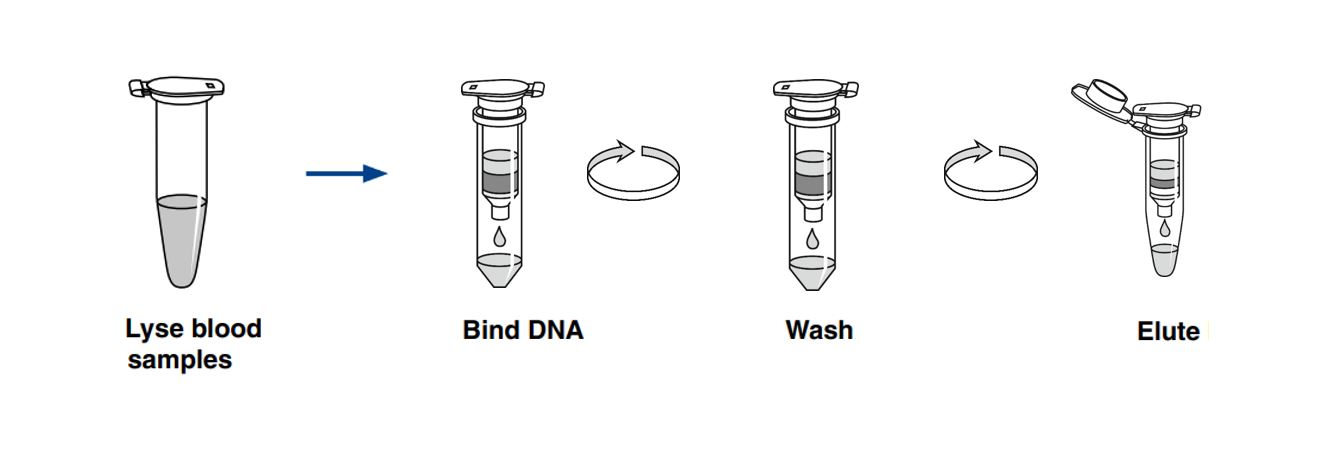
NucleoSpin DX Blood - Procedure
gDNA from whole blood samples– for in vitro diagnostic purposes
The NucleoSpin Dx Blood kit is a generic system for the isolation and purification of genomic DNA from human whole blood samples for subsequent in vitro diagnostic purposes. The kit can be used with fresh and frozen human whole blood treated with EDTA, citrate, or heparin, from common blood collection systems. The kit is designed to be used with any downstream application employing enzymatic amplification and detection of DNA (e.g., PCR).
The NucleoSpin Dx Blood procedure is based on the proven MACHEREY-NAGEL silica membrane technology. First, the blood samples are lysed in the presence of chaotropic salts and Proteinase K. The genomic DNA in the lysate is then bound to a NucleoSpin Dx Blood Column. Subsequently, the membrane with bound nucleic acids is washed and finally highly pure genomic DNA is eluted.
Excellent DNA recovery
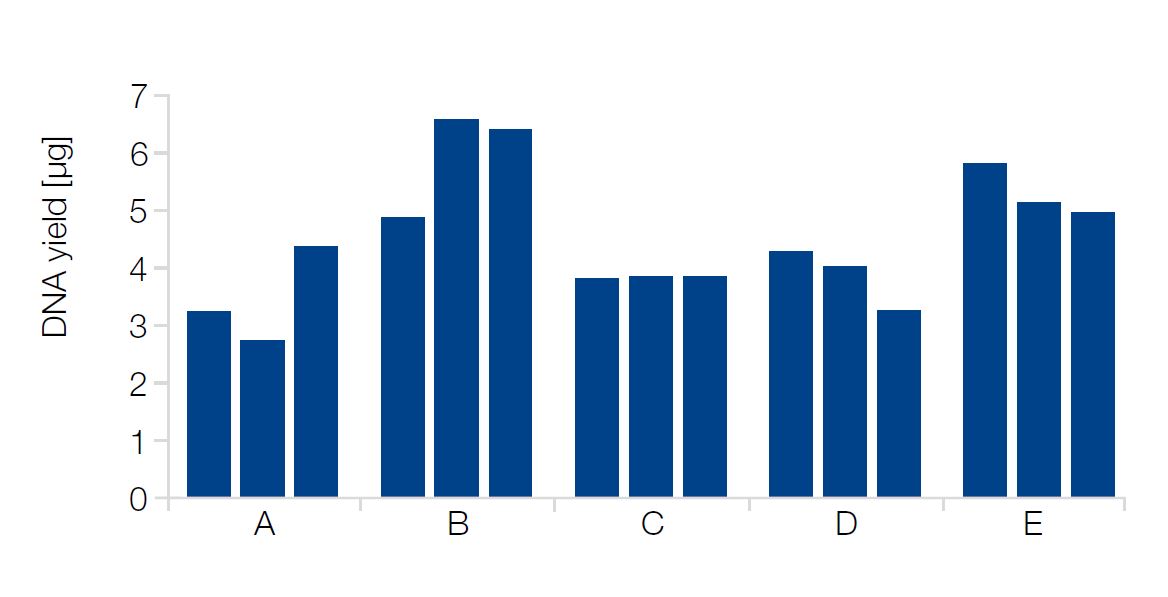
DNA was isolated from triplicates of blood samples
(200 μL, EDTA) from 5 individuals (A–E).
The DNA yield is 2.7–6.6 μg, depending on blood sample.
Reliable performance regardless the anticoagulant used

DNA was isolated from 15 individual blood samples, stabilized with different anticoagulants: EDTA, citrate, and heparin.
All samples are showing reliable good performance of
qPCR.
Selection of suitable blood collection systems
| Blood collecting system | Manufacturer |
|---|---|
| S-Monovette Li-Heparin | Sarstedt |
| S-Monovette EDTA | Sarstedt |
| S-Monovette Citrat | Sarstedt |
| K3 EDTA | DELTA LAB |
| BD VACUTAINER K2E | BD |
| VACUETTE EDTA | Greiner Bio-One |
| K2 EDTA | APTACA |
NucleoSpin Dx Blood is compatible with common blood collection tubes and anticoagulants.
It is suitable for EDTA, citrate, and heparin blood collecting systems and for fresh and frozen blood samples.
A selection of suitable blood collection devices is shown.
 NucleoSpin Dx Virus, Mini kit for CE certified purification of viral RNA/DNA
NucleoSpin Dx Virus, Mini kit for CE certified purification of viral RNA/DNA